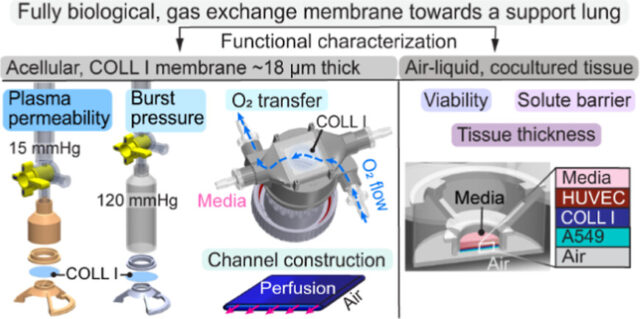
A means of long-term respiratory support is needed for the nearly one million chronic lung disease patients hospitalized annually. Extracorporeal membrane oxygenation can support patients for months, but clot formation within oxygenators and bleeding complications make it infeasible for permanent support. An endothelial cell coating on these devices could leverage cells’ ability to reduce clot initiation and propagation, but long-term binding to artificial materials has not been achieved. The goal of these studies was to engineer a preliminary, fully biological tissue that mimics the alveolar-capillary barrier and could function as the gas-exchange membrane of an implantable, biofabricated support lung for years. High-concentration, type I collagen membranes were made to be 18.8 ± 3.6 μm-thick and characterized in terms of mechanical strength, water permeability, and oxygen transfer under static, air–liquid conditions. The membranes were cocultured with human umbilical vein endothelial cells (HUVECs) and A549 lung epithelial cells on opposing sides to evaluate tissue viability in air–liquid conditions and permeability to the albumin mimic, 70 kDa-FITC dextran. The 18.8 ± 3.6 μm-thick acellular collagen I hydrogel withstood ≥120 mmHg and transferred 2.16 ± 0.5 μL/cm2/mmHg/h of plasma. It was oxygen permeable and produced 75% of the gas transfer of a 51 μm, implantable, silicone sheet. Cell cocultures remained viable in air–liquid conditions, and dextran permeability emphasized the need to include an alveolar epithelium to improve barrier function. Lastly, the method was expanded into casting a parallel-plate, perfusable channel as the functional, representative element of a booster lung. The channel provided a 16.8 ± 3.4 μm diffusion distance across 4 cm2 of surface area and maintained an air–liquid interface. Future work should examine cross-linking the collagen I for equally strong but thinner membranes (≤10 μm), transition to induced pluripotent stem cell cultures, and implement multichannel casting to increase the surface area needed for a biofabricated, intracorporeal, support lung.
- Journal:
- ACS Biomaterials Science & Engineering
- Year:
- 2026
- PMID:
- 41968579
- DOI:
- 10.1021/acsbiomaterials.6c00046

